Overview
What are normal calcium concentrations and requirements?
Serum concentration
2.2-2.6 mmol/L
Requirements
0.1 mmol/kg/day
What are the functions of calcium in the body?
- Bone formation and metabolism:
- Contributes to strength and structure
- Role as a strong cation in acid-base balance
- Coagulation of blood (cofactor in coagulation pathway)
- Cellular functions:
- Excitation–contraction coupling in cardiac, skeletal and smooth muscle
- Cardiac action potentials and pacemaker activity
- Regulation of cell growth and apoptosis
- Cofactor for many enzymes (e.g. lipase) and proteins
- Membrane integrity & permeability
- Ciliary motility
- Cellular communication:
- Intracellular secondary messenger systems
- Secretory processes:
- Release of neurotransmitters
- Hormone release
- Catecholamine responsiveness
What is the role of calcium in enzyme activation?
- The protein calmodulin activates many enzymes in the presence of calcium:
- Calcium-calmodulin complexes bind to enzymes acting as a co-factor
- Known as a ‘calcium-binding regulatory protein’
- Enzymes activated by calmodulin include:
- Glycogen synthase
- Adenyl cyclase
- Pancreatic lipase
- Pyruvate carboxylase
- Phospholipase A2
Why is calcium required for clotting?
- Ionized calcium is essential for many stages of both coagulation and platelet plug formation
- Multiple roles in coagulation pathway (Calcium is known as Factor IV):
- Required for the activation of factors VII, IX and X
- Acts as a co-factor in the conversion of prothrombin (II) to thrombin (IIa) – binds to the tenase enzyme where it acts as a catalyst speeding up the reaction
- Important in platelet activation and aggregation:
- Increase in intracellular calcium upon stimulation activates a number of calcium-dependent enzymes and regulated release of active substances
- Standard anticoagulant solutions contain citrate which chelates calcium ions and efficiently prevents clotting
What role does calcium play in cell excitability?
- Excitability of neuronal and cardiac tissue is inversely proportional to extracellular calcium levels
- Due to the direct effect calcium plays in binding to voltage-gated sodium and potassium channels and slowing their opening
- Results in altered membrane threshold potentials
- Low calcium levels cause increased membrane excitability:
- Reduces the membrane threshold potential, thus narrowing the gap between the resting and threshold potentials
- Can result in arrhythmias, tetany and seizures
- High calcium levels cause decreased membrane excitability:
- Reduces the membrane threshold potential, thus widening the gap between the resting and threshold potentials
- Is used in hyperkalaemia (through calcium administration) to restore a normal gradient between the membrane threshold potential and pathologically elevated resting membrane potential
Body & Plasma Stores
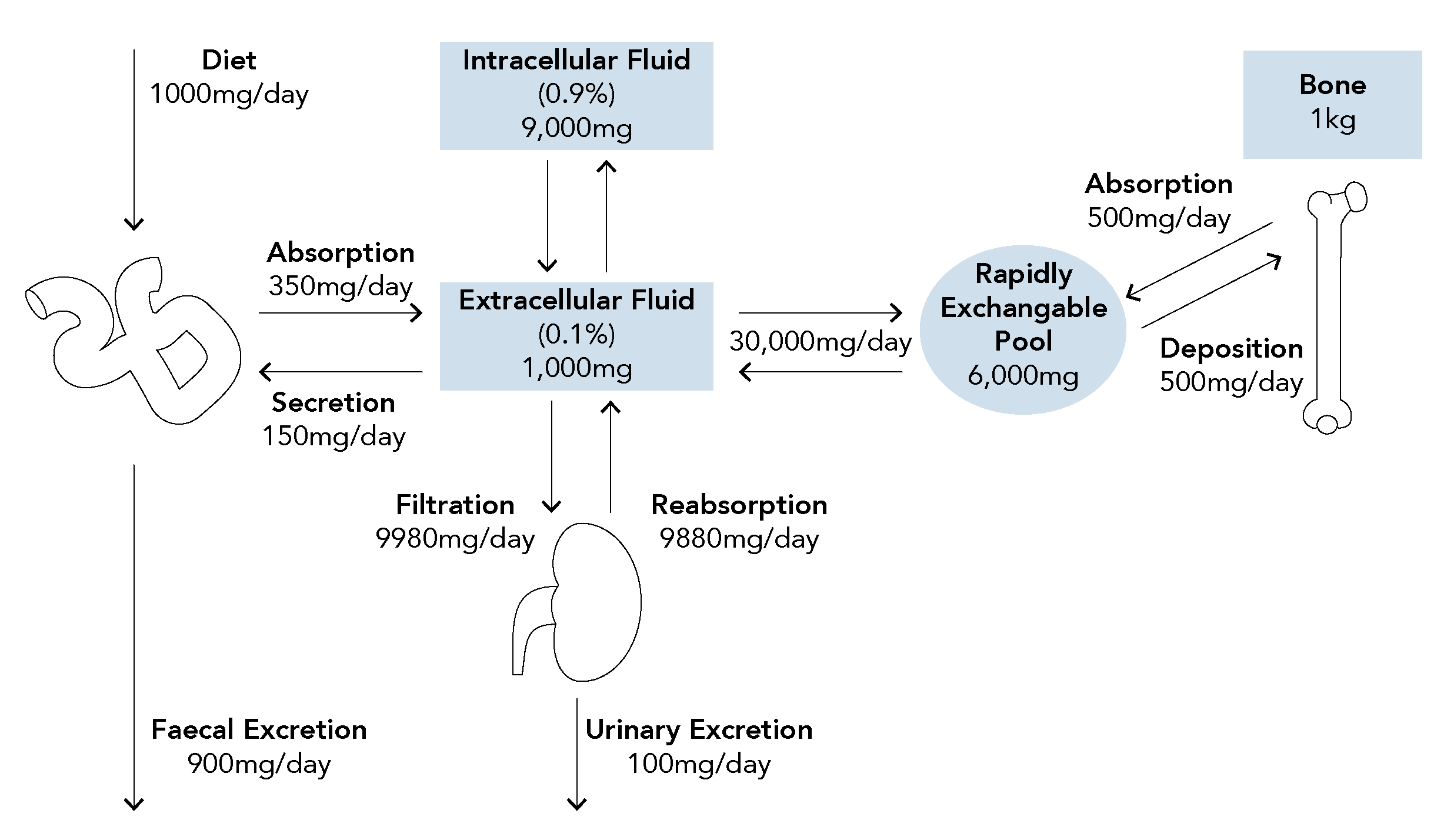
How is calcium stored in the body?
What is the normal daily turnover of calcium?
What is the normal concentration of calcium in the blood and what forms is it found in?
- Biological effect is determined by the amount of ionised calcium which is under tight homeostatic control and usually remains constant
How are calcium levels affected by changes in albumin concentration?
- Total calcium levels are generally dependent on the level of plasma albumin
- With reduced serum albumin proportionally less calcium is bound
- This results in a lower measured total plasma calcium
How can the effect of albumin levels be accounted for?
- A corrective formula (Payne’s formula) is commonly applied to account for albumin concentrations when considering the measured total calcium levels
- A correction factor of 0.02 mmol/L is added to the measured calcium level for every 1 g/L of albumin below a value of 40 g/L)
- Results in the formula:
Corrected Ca2+ = measured Ca2+ + 0.02 x (40 – serum albumin)
Corrected Calcium
- For example, if the measured total plasma calcium is 1.876 mmol/L and plasma albumin is 26 g/L:
| Corrected Ca2+ | = 1.83 + 0.02 x (40 – 26) = 1.83 + 0.28 = 2.11 mmol/L |
Worked Example
Which factors affects serum calcium levels?
- Ca2+ ions competes for binding sites on albumin with H+ ions
- As pH changes the proportion of calcium bound to albumin and freely ionised in the plasma varies
- For every 0.1 unit reduction in plasma pH, the albumin bound calcium decreases by 0.07 mmol/L and ionised calcium increases by 0.07 mmol/L
- Hyperventilation can lead to alkalosis, and thus reduced plasma ionised calcium:
- As ionised calcium determines clinical effects tetany may be precipitated
Regulation
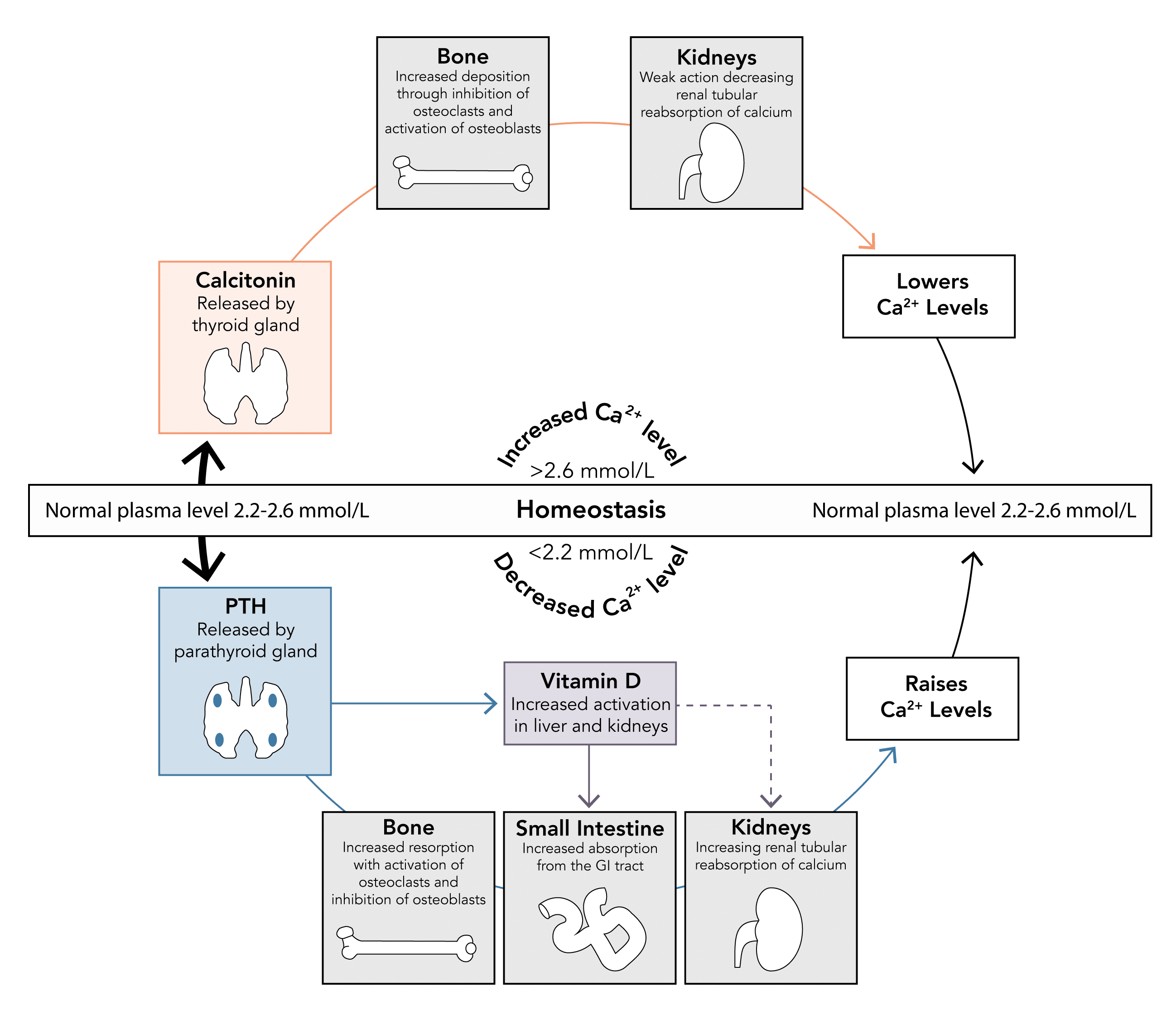
How is calcium homeostasis maintained?
- Calcium homeostasis can be considered through the action of:
- Three organs: bone, kidney, small intestine
- Three hormones: parathyroid hormones, calcitonin, vitamin D metabolites
Which hormones are involved in calcium homeostasis and what are their roles?
Hormone
Production & Secretion
Action
Parathyroid Hormone
- Protein hormone produced by chief cells of the parathyroid gland
- Secretion stimulated by a decrease in serum calcium levels
- Normal levels are 1.3 – 6.8 pmol/L
- Acts to increase serum calcium and decrease serum phosphate levels through a number of mechanisms:
- Kidney: Increases calcium reabsorption
- Bone: Mobilises calcium from bone stores by stimulating osteoclast activity and inhibiting osteoblast activity
- GI Tract: Increases GI absorption of calcium and phosphate indirectly by production active vitamin D metabolites
Vitamin D3
(cholecalciferol)
(cholecalciferol)
- Originates from:
- Dietary intake
- Synthesis in the skin through effects of UV light on 7-dehydrocholesterol
- Modified by liver and kidney to form active 1-25-hydroxycholecalciferol - under the influence of parathyroid hormone
- Levels also increase in response to low phosphate levels
- Acts to increase serum calcium levels
- GI Tract: Increases GI absorption by upregulating number and activity of transport proteins
- Kidneys: Weak effect to increase calcium and phosphate reabsorption
Calcitonin
(minimal role)
(minimal role)
- Peptide hormone produced by parafollicular cells of the thyroid gland
- Secretion stimulated by an increase in serum calcium levels
- Plays a minimal role in calcium homeostasis, secreted only when levels rise above normal limits
- Acts to decrease serum calcium levels when elevated (physiological antagonist to PTH)
- Bone: Prevents resorption by inhibiting osteoclast activity and stimulating osteoblast activity
- Kidneys: Weak action to decrease calcium reabsorption